As part of the Personal Genome Project (PGP), my genome was recently sequenced by Complete Genomics. My PGP profile, including the sequence, is here, and their report on my genome is here. As I play around with the best ways to analyze these data, I’ll write additional posts, but for now I’ve noticed only one thing: I’m almost surprised by how unsurprising my full genome sequence is.
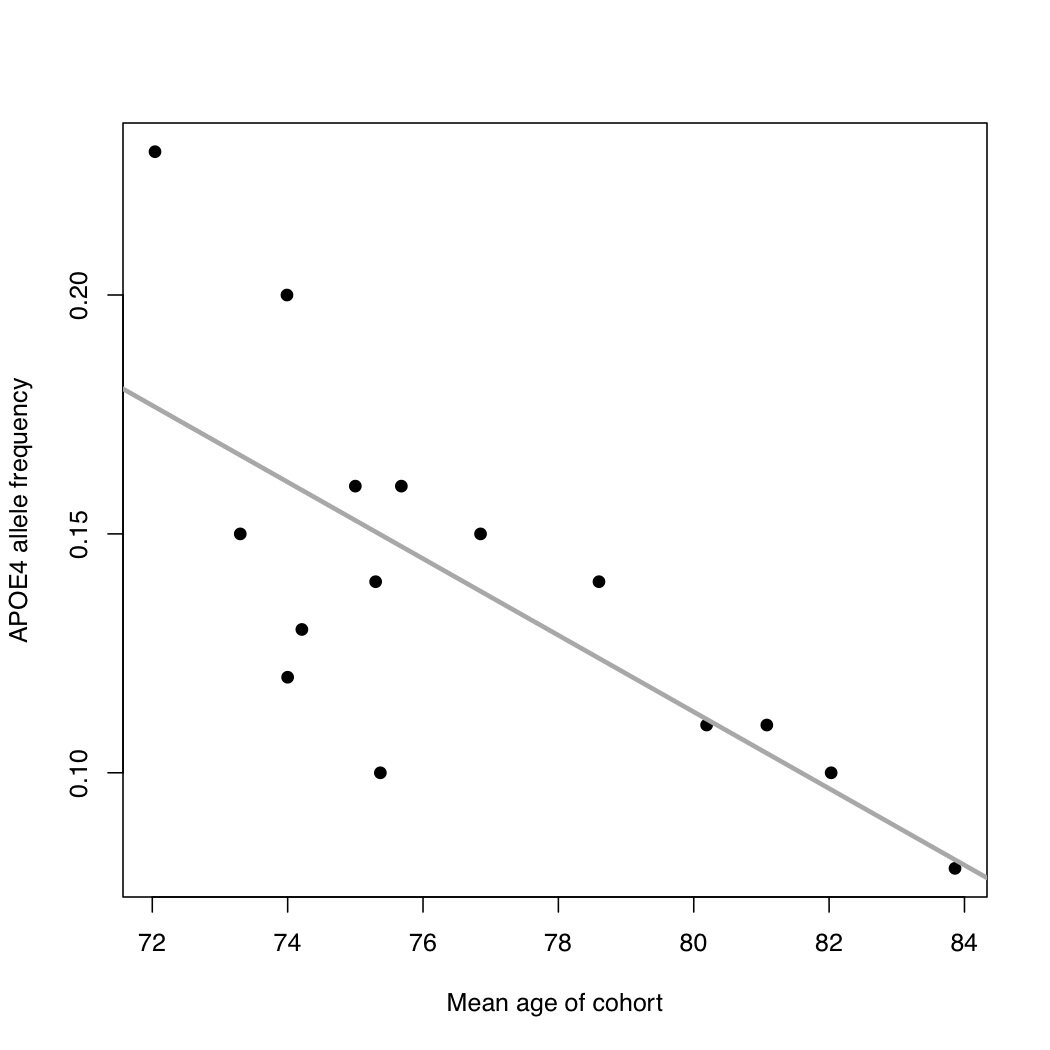
According the the PGP’s genome annotator, I have two variants of “high” clinical relevance. The first is the APOE4 allele, which Luke had already reported that I carry. The second is a variant that causes alpha-1-antitrypsin deficiency, which is also typed by 23andMe.
Of course, this is all quite reassuring. Long-time readers will remember that last year I was briefly worried that I might have Brugada syndrome. I do not carry any of the known pathogenic mutations (modulo worries about false negatives); this of course is now unsurprising, but would have been really nice information to have, say, when I was talking with a cardiologist last year.
 Out in Nature this week is a paper by three Genomes Unzipped authors reporting 71 new genetic associations with inflammatory bowel disease (IBD). This breaks the record for the largest number of associations for any common disease, and includes many new and interesting biological insights that you should all go and read about in the paper itself (pay-to-access I’m afraid) or on the Sanger Institute’s website.
Out in Nature this week is a paper by three Genomes Unzipped authors reporting 71 new genetic associations with inflammatory bowel disease (IBD). This breaks the record for the largest number of associations for any common disease, and includes many new and interesting biological insights that you should all go and read about in the paper itself (pay-to-access I’m afraid) or on the Sanger Institute’s website.
 I have no strong family history of any disease, despite having 7 blood aunts and uncles and countless cousins. So when I sent my spit off to 23andMe at the start of the Genomes Unzipped project, I was expecting something very similar to
I have no strong family history of any disease, despite having 7 blood aunts and uncles and countless cousins. So when I sent my spit off to 23andMe at the start of the Genomes Unzipped project, I was expecting something very similar to 
 RSS
RSS Twitter
Twitter
Recent Comments